Patiromer
 | |
| Clinical data | |
|---|---|
| Trade names | Veltassa |
| Synonyms | RLY5016 |
AHFS/Drugs.com | veltassa |
| Routes of administration | Oral suspension |
| ATC code |
|
| Legal status | |
| Legal status |
|
Pharmacokinetic data | |
| Bioavailability | Not absorbed |
| Metabolism | None |
| Onset of action | 7 hrs |
| Duration of action | 24 hrs |
| Excretion | Feces |
| Identifiers | |
IUPAC name
| |
| CAS Number |
|
PubChem SID |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL |
|
| Chemical and physical data | |
| Formula | [(C3H3FO2)182·(C10H10)8·(C8H14)10]n [Ca91(C3H2FO2)182·(C10H10)8·(C8H14)10]n (calcium salt) |
Patiromer (USAN, trade name Veltassa) is a drug used for the treatment of hyperkalemia (elevated blood potassium levels), a condition that may lead to palpitations and arrhythmia (irregular heartbeat). It works by binding potassium in the gut.[1][2]
Contents
1 Medical uses
2 Adverse effects
3 Interactions
4 Pharmacology
4.1 Mechanism of action
4.2 Pharmacokinetics
5 Chemistry
6 History
6.1 Studies
6.2 Approval
7 See also
8 References
Medical uses
Patiromer is used for the treatment of hyperkalemia, but not as an emergency treatment for life-threatening hyperkalemia, because it acts relatively slowly.[2] Such a condition needs other kinds of treatment, for example calcium infusions, insulin plus glucose infusions, salbutamol inhalation, and hemodialysis.[3]
Typical reasons for hyperkalemia are renal insufficiency and application of drugs that inhibit the renin–angiotensin–aldosterone system (RAAS) – e.g. ACE inhibitors, angiotensin II receptor antagonists, or potassium-sparing diuretics – or that interfere with renal function in general, such as nonsteroidal anti-inflammatory drugs (NSAIDs).[4][5]
Adverse effects
Patiromer was generally well tolerated in studies. Side effects that occurred in more than 2% of patients included in clinical trials were mainly gastro-intestinal problems such as constipation, diarrhea, nausea, and flatulence, and also hypomagnesemia (low levels of magnesium in the blood) in 5% of patients, because patiromer binds magnesium in the gut as well.[2][6]
Interactions
Patiromer was tested for drug-drug interactions with 28 drugs and showed binding or interaction with 14 of these drugs. This could reduce their availability and thus effectiveness,[2] wherefore patiromer has received a boxed warning by the US Food and Drug Administration (FDA), telling patients to wait for at least six hours between taking patiromer and any other oral drugs.[7]
Of the 14 drugs that did show an interaction in vitro, 12 were selected for further testing in phase 1 studies in healthy volunteers to assess whether the results seen in vitro translated into an effect in people. These studies showed patiromer did not alter the absorption of nine of the 12 drugs when co-administered. Patiromer reduced absorption of three drugs when co-administered, however, there was no interaction when patiromer and these three drugs were taken 3 hours apart.[8]
This information was submitted to the FDA in the form of a supplemental New Drug Application (sNDA) and as a result, in November 2016 the FDA approved the removal of the boxed warning regarding the separation of patiromer and other oral medications. The updated label recommends patients take patiromer at least 3 hours before or 3 hours after other oral medications.[9]
Pharmacology
Mechanism of action
Patiromer works by binding free potassium ions in the gastrointestinal tract and releasing calcium ions for exchange, thus lowering the amount of potassium available for absorption into the bloodstream and increasing the amount that is excreted via the feces. The net effect is a reduction of potassium levels in the blood serum.[2][4]
Lowering of potassium levels is detectable 7 hours after administration. Levels continue to decrease for at least 48 hours if treatment is continued, and remain stable for 24 hours after administration of the last dose. After this, potassium levels start to rise again over a period of at least four days.[2]
Pharmacokinetics
Patiromer is not absorbed from the gut, is not metabolized, and is excreted in unchanged form with the feces.[2]
Chemistry
The substance is a cross-linked polymer of 2-fluoroacrylic acid (91% in terms of amount of substance) with divinylbenzenes (8%) and 1,7-octadiene (1%). It is used in form of its calcium salt (ratio 2:1) and with sorbitol (one molecule per two calcium ions or four fluoroacrylic acid units), a combination called patiromer sorbitex calcium.[10]
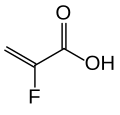
2-fluoroacrylic acid
o-divinylbenzene
p-divinylbenzene

1,7-octadiene
Patiromer sorbitex calcium is an off-white to light brown, amorphous, free-flowing powder. It is insoluble in water, 0.1 M hydrochloric acid, heptane, and methanol.[2][10]
History
Studies
In a Phase III multicenter clinical trial including 237 patients with hyperkalemia under RAAS inhibitor treatment, 76% of participants reached normal serum potassium levels within four weeks. After subsequent randomization of 107 responders into a group receiving continued patiromer treatment and a placebo group, re-occurrence of hyperkalemia was 15% versus 60%, respectively.[11]
Approval
The US FDA approved patiromer in October 2015.[7] The drug is now also approved in Europe as of July 2017[update].
See also
ZS-9, another potassium binder
References
^ Henneman, A; Guirguis, E; Grace, Y; Patel, D; Shah, B (2016). "Emerging therapies for the management of chronic hyperkalemia in the ambulatory care setting". American Journal of Health-System Pharmacy. 73 (2): 33–44. doi:10.2146/ajhp150457. PMID 26721532..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ abcdefgh FDA Professional Drug Information for Veltassa.
^ Vanden Hoek TL, Morrison LJ, Shuster M, Donnino M, Sinz E, Lavonas EJ, Jeejeebhoy FM, Gabrielli A; Morrison; Shuster; Donnino; Sinz; Lavonas; Jeejeebhoy; Gabrielli (2010-11-02). "Part 12: cardiac arrest in special situations: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care". Circulation. 122 (18 Suppl 3): S829–61. doi:10.1161/CIRCULATIONAHA.110.971069. PMID 20956228.CS1 maint: Multiple names: authors list (link)
^ ab Esteras, R.; Perez-Gomez, M. V.; Rodriguez-Osorio, L.; Ortiz, A.; Fernandez-Fernandez, B. (2015). "Combination use of medicines from two classes of renin-angiotensin system blocking agents: Risk of hyperkalemia, hypotension, and impaired renal function". Therapeutic Advances in Drug Safety. 6 (4): 166–76. doi:10.1177/2042098615589905. PMC 4530349. PMID 26301070.
^ Rastegar, A; Soleimani, M (2001). "Hypokalaemia and hyperkalaemia". Postgraduate Medical Journal. 77 (914): 759–64. doi:10.1136/pmj.77.914.759. PMC 1742191. PMID 11723313.
^ Tamargo, J; Caballero, R; Delpón, E (2014). "New drugs for the treatment of hyperkalemia in patients treated with renin-angiotensin-aldosterone system inhibitors -- hype or hope?". Discovery medicine. 18 (100): 249–54. PMID 25425465.
^ ab "FDA approves new drug to treat hyperkalemia". FDA. 21 October 2015.
^ Pharmabiz: US FDA approves removal of boxed warning on Relypsa's hyperkalemia drug, Veltassa.
^ FDA Package Insert: VELTASSA (patiromer) for oral suspension.
^ ab RxList: Veltassa.
^ Weir, Matthew R.; Bakris, George L.; Bushinsky, David A.; Mayo, Martha R.; Garza, Dahlia; Stasiv, Yuri; Wittes, Janet; Christ-Schmidt, Heidi; Berman, Lance; Pitt, Bertram (2015). "Patiromer in Patients with Kidney Disease and Hyperkalemia Receiving RAAS Inhibitors". New England Journal of Medicine. 372 (3): 211–21. doi:10.1056/NEJMoa1410853. PMID 25415805.

